 
Interactive approach establishes a well-deserved academic connect between you and Master Teachers. Sessions get recorded for you to access for quick revision later, just by a quick login to your account. Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts. 
WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. Gamma rays, E-beam, and X-rays each have their set of advantages and disadvantages when it comes to irradiation modes. But due to increasing costs, other alternatives are also being seen such as E-beam technology which has gained popularity but is not suitable for many products. When it undergoes beta-decay it reaches the excited state of Nickel-60.Ĭobalt-60 is the longest living radioactive isotope of Cobalt which is primarily used as a radiation source in irradiation and sterilization processes. The diagram below depicts the energy and frequency of the gamma radiation emitted by Cobalt-60 to expend the extra energy it possesses. In the absence of cobalt-60, 4-MV photon beams can be used also which are adequate but the concern over 6-MV photons does not compromise treatment and the treatment planning advantages outweigh the small differences in beam properties. There is a concern while using 6-MV photons that they have a larger buildup region and can underdose superficial tissues as most radiation oncology centers have a linear accelerator only. Intensity-modulated radiation therapy or IMRT is being increasingly used in the treatment of advanced diseases in which a linear beam accelerator delivers low-energy photons. These are operated either continuously or in large batches.Ĭobalt 60 is often the preferred mode of radiation in the treatment of laryngeal cancer. The industrial Cobalt-60 is used for phytosanitary applications and medical device sterilization as well. The radiation from a Cobalt source has been primarily used as a bactericide in the treatment of commodities such as spices, dry fruits, fruits, poultry, onions, and potatoes. 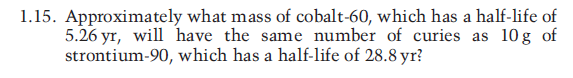
The number of Cobalt 60 protons neutrons electrons are 27,33, and 27 respectively. The -decay energy is low and shielded easily while the gamma rays have high energy emission lines around 1.3MeV and are highly penetrating. Cobalt 60 emits high-intensity gamma rays. All isotopes of an element have the same atomic number but different atomic mass.Ĭobalt mass is 58.993 and the mass of Cobalt isotopes varies from 50u to 73u. Isotopes: Isotopes are elements with the same number of protons but they differ in the number of neutrons. It has the longest half-life among all the Cobalt isotopes. It is essential as it provides information about how quickly unstable atoms undergo radioactive decay and how long they survive.Ĭobalt 60 half-life is 5.2713 years which is one of its main advantages being a high-intensity gamma-ray emitter. Half-Life: half-life is a commonly used term in nuclear physics which depicts the amount of time required for a quantity of element to reduce to half of its initial value. It is produced by nuclear bombardment of stable Cobalt in a nuclear reactor Properties of Cobalt-60Ĭobalt 60 is a common source of radiation that emits gamma rays or an accelerator producing beam of electrons. It is produced artificially in nuclear reactors. It is a radioactive isotope of Cobalt commonly used in industries and medicines. Most cobalt isotopes have a half-life of less than 1 second and the most stable isotope of Cobalt is Cobalt 60 with a half-life of 5.2713 years. Other than the stable isotope Co 59, there are a total of 28 radioactive cobalt isotopes found on earth. The atomic mass of Cobalt is 58.993 and its atomic number is 27. It is obtained by reductive smelting of its ore to produce a hard, lustrous, and silver-grey metal. It is a naturally occurring element found in the Earth’s crust in the form of various ores. Cobalt is an element present in group 9 of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
